 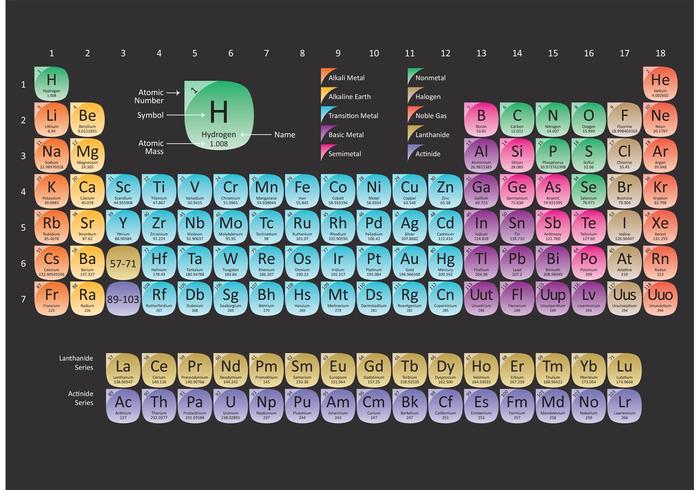
The molar mass of a compound is the mass in grams of 1 mole of that substance, a property expressed in units of grams per mole (g/mol)īecause the definitions of both the mole and the formula mass are based on the same reference substance, 12C, the molar mass of any compound is numerically equivalent to its formula weight in amu. 15 Nitrogen 7 16 Oxygen 8 17 Fluorine 9 4.00 - Neon not round them further when performing calculations. The masses of 1 mole of different compunds, however, are different, since the masses of the individual molecules are drastically different. Weve discussed mass at length by doing molar mass conversions. Similar to atoms, 1 mole of any compound contains the same number of molecules as 1 mole of any other compund. The given mass of K (4. 94 of these occur naturally, elements of higher atomic numbers were only produced by humans and are referred to as synthetic substances.\). Referring to the periodic table, the atomic mass of K is 39.10 amu, and so its molar mass is 39.10 g/mol. Today, 118 elements are known – ascending from hydrogen to oganesson. Include appropriate units for molar mass. Use the Periodic Table from class and the appropriate significant figures from the table. Give the molar mass of the compound below. Enter a chemical formula to calculate its molar mass (e.g. The structure of the periodic table in seven horizontal periods and 18 vertical periods allows the ordering of chemical elements based on elementary properties, such as their increasing atomic number, similarity, reactivity or preferred bonding partners. Include appropriate units for molar mass. Many then unknown elements could be correctly predicted by their similarity to already known chemical elements. The periodic table was developed in 1869 by the Russian Dmitri Mendeleev and independently a few months later by the German Lothar Meyer. When the chemical elements are thus arranged, there is a recurring pattern called the periodic law in their properties, in which elements in the same column (group. Every chemical element has a specific atomic number, which provides insight into the number of protons present within its nucleus. The position of an element immediately reveals its essential chemical characteristics, elements with similar properties are listed one below the other. periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number i.e., the total number of protons in the atomic nucleus. The periodic table lists the atomic mass of carbon as 12.011 amu the average molar mass of carbonthe mass of 6.022 × 10 23 carbon atomsis therefore 12.011 g/mol: Substance (formula) Atomic, Molecular, or Formula Mass (amu) Molar Mass (g/mol) carbon (C) 12.011 (atomic mass) 12.011: ethanol (C 2 H 5 OH) 46. For a fully interactive version with orbitals, isotopes, compounds, and free printouts, visit. The lightest chemical element is Hydrogen and the heaviest is Hassium. Design Copyright © 2017 Michael Dayah (). The table is divided into rows (periods) and columns (groups). For chemistry students and teachers: The tabular chart on the right is arranged by Atomic mass (weight). Visualize trends, 3D orbitals, isotopes, and mix compounds. I also usually use the full number given on the periodic table just to be exact as well, but I have noticed some people round their masses from the periodic table to about 2 or 3 decimal places. In the periodic table of elements, all known chemical elements are arranged according to increasing nuclear charge (atomic number). Interactive periodic table showing names, electrons, and oxidation states. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
